Bard is currently experiencing many hundred lawsuits by individuals who've had the Restoration and G2 IVC filters inserted, resulting in injury. Plaintiffs claim that the organization promoted poorly developed devices and didn't warn the medical community and community about possible pitfalls.
The initial lawsuits concerning the Bard devices were submitted in 2012 in Missouri and California state courts. In October 2014, federal lawsuits were combined into a multidistrict litigation (MDL) in the U.S. District Court for your Southern District of Indiana, overseen by U.S. District Judge Richard M. Small.
Side Effects From Inferior Vena Cava
The very first scenario to go to test was resolved in January of 2015. After his Bard Restoration IVC filter smashed apart, causing a little steel “leg” from the product to journey to his heart Kevin Philips suffered significant medical harm. The steel fragment perforated his heart, necessitating not close - a restoration period that was debilitating plus heart surgery. The case was satisfied for an undisclosed amount.
Bard started receiving claims regarding IVC filter system malfunctioning as early as 2004. They did not reveal this information towards the Food or even to the medical group but instead continued marketing the products although the organization understood about product failures.
Plaintiffs who've been harmed by way of a faulty medical unit could be entitled to real problems (medical fees, missing salaries, discomfort, and suffering) and a few could be qualified to receive punitive damages (to punish the company) if it can be proven the manufacturer believed about disorders in their goods and continued to market them any way.
IVC Filter Lawsuits Moving Forward
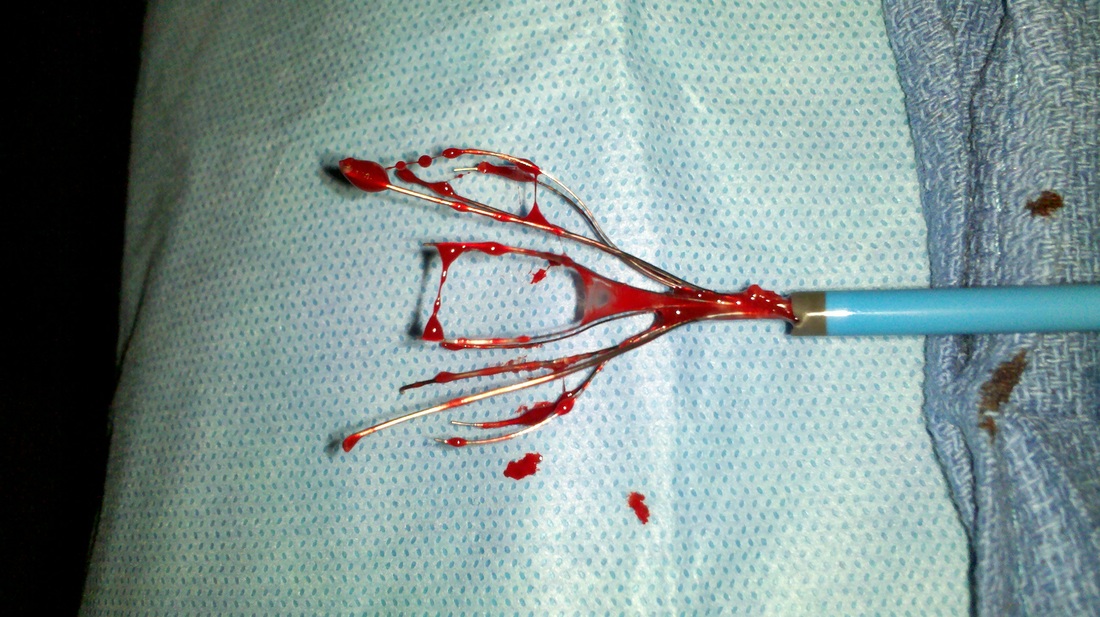
IVC filter suit infographic An Inferior Vena Cava (IVC) filter can be a medical unit the lungs. IVC filters are implanted the greatest vein in the body, to the Vena Cava, which conveys blood that was “used” from areas and the tissues to the heart, where it's excited to the lungs to be re-used and oxygenated. IVC filters are utilized in patients with certain clotting disorders like deep-vein thrombosis (DVT) who can't take anticoagulant treatment. Be a pulmonary embolism which may be deadly and they're meant to “catch” blood clots before they attain the lungs.
Bard, previously known as C.R. Bard, is product maker situated with an estimated yearly income of approximately $3 billion, in Nj. The company produces a number of medical products for medical, urological. After it was withdrawn in the marketplace, Bard currently manufacturers the G2 IVC filter that has been intended to replace a previous device, the Restoration IVC filter.
IVC filters were made to protect patients from injury but unfortunately, many gadgets including the Restoration and G2 IVC filters have not performed as planned. The products have now been shown to cause severe complications due to flaws.
Additionally, although Food has stated that IVC filters ought to be removed as soon as the danger of blood clots has transferred, many products have already been left in position for a lot longer and retrieval has proven impossible or difficult.
Numerous lawsuits have now been filed against Bard regarding harm caused by the Healing. The maker has also faced tens of thousands of lawsuits for additional faulty medical units such as a transvaginal mesh.
The initial lawsuits concerning the Bard devices were submitted in 2012 in Missouri and California state courts. In October 2014, federal lawsuits were combined into a multidistrict litigation (MDL) in the U.S. District Court for your Southern District of Indiana, overseen by U.S. District Judge Richard M. Small.
Side Effects From Inferior Vena Cava
The very first scenario to go to test was resolved in January of 2015. After his Bard Restoration IVC filter smashed apart, causing a little steel “leg” from the product to journey to his heart Kevin Philips suffered significant medical harm. The steel fragment perforated his heart, necessitating not close - a restoration period that was debilitating plus heart surgery. The case was satisfied for an undisclosed amount.
Bard started receiving claims regarding IVC filter system malfunctioning as early as 2004. They did not reveal this information towards the Food or even to the medical group but instead continued marketing the products although the organization understood about product failures.
Plaintiffs who've been harmed by way of a faulty medical unit could be entitled to real problems (medical fees, missing salaries, discomfort, and suffering) and a few could be qualified to receive punitive damages (to punish the company) if it can be proven the manufacturer believed about disorders in their goods and continued to market them any way.
IVC Filter Lawsuits Moving Forward
IVC filter suit infographic An Inferior Vena Cava (IVC) filter can be a medical unit the lungs. IVC filters are implanted the greatest vein in the body, to the Vena Cava, which conveys blood that was “used” from areas and the tissues to the heart, where it's excited to the lungs to be re-used and oxygenated. IVC filters are utilized in patients with certain clotting disorders like deep-vein thrombosis (DVT) who can't take anticoagulant treatment. Be a pulmonary embolism which may be deadly and they're meant to “catch” blood clots before they attain the lungs.
Bard, previously known as C.R. Bard, is product maker situated with an estimated yearly income of approximately $3 billion, in Nj. The company produces a number of medical products for medical, urological. After it was withdrawn in the marketplace, Bard currently manufacturers the G2 IVC filter that has been intended to replace a previous device, the Restoration IVC filter.
IVC filters were made to protect patients from injury but unfortunately, many gadgets including the Restoration and G2 IVC filters have not performed as planned. The products have now been shown to cause severe complications due to flaws.
Additionally, although Food has stated that IVC filters ought to be removed as soon as the danger of blood clots has transferred, many products have already been left in position for a lot longer and retrieval has proven impossible or difficult.
Numerous lawsuits have now been filed against Bard regarding harm caused by the Healing. The maker has also faced tens of thousands of lawsuits for additional faulty medical units such as a transvaginal mesh.